
Author: Didier Tranchier (Ph.D.)
🔍 Artificial Intelligence: A New Pharmaceutical Prescription Model – English version
Artificial intelligence (AI), specifically large language models (LLMs), has established itself in just a few years as one of the most transformative technologies of our time. While its impact on all economic sectors is already well documented, its influence on medical practices—and more specifically on the pharmaceutical prescribing model—remains insufficiently analyzed. This is precisely the focus of this article: to examine how AI is giving rise to a new prescribing model—one that is more powerful than the traditional model but raises a fundamental question of sovereignty that pharmaceutical companies can no longer ignore.
To fully grasp the scope of this transformation, we must first examine the structural weaknesses of the model it is replacing.
-
A physical prescription model in sharp decline
Let us first clarify what we mean by the “physical prescription model” for pharmaceutical products: this refers to all activities aimed at informing physicians about pharmaceutical products through in-person interactions and physical materials, with the goal of increasing their prescription rates. This model relies on several key functions within pharmaceutical companies:
- The sales teams who visit healthcare professionals, hospitals, and pharmacies directly
- Medical medical affairs that disseminate scientific and medical information to healthcare professionals individually or collectively, particularly at conferences, and promote projects with clinical or therapeutic objectives, particularly research projects
- The marketing, which produces content and organizes campaigns targeting the entire ecosystem
The effectiveness of this model has long been called into question. For example, a review of the scientific literature published as early as 2010 highlights the lack of demonstrated systematic effectiveness.
Furthermore, the growing and ongoing restrictions on access to doctors have undermined the effectiveness of this model while driving up its costs. The rapid development of digital tools has not reversed this trend: although it has introduced new channels of interaction, it has not restored the model’s effectiveness.
In this context, the advent of artificial intelligence does not merely undermine this model; it renders it obsolete, creating a new, radically different model.
-
AI is already the primary reference tool for doctors and patients
Since the launch of ChatGPT 3.5 on November 30, 2022, the adoption of these tools has seen unprecedented growth, as illustrated in Figure 1 below:
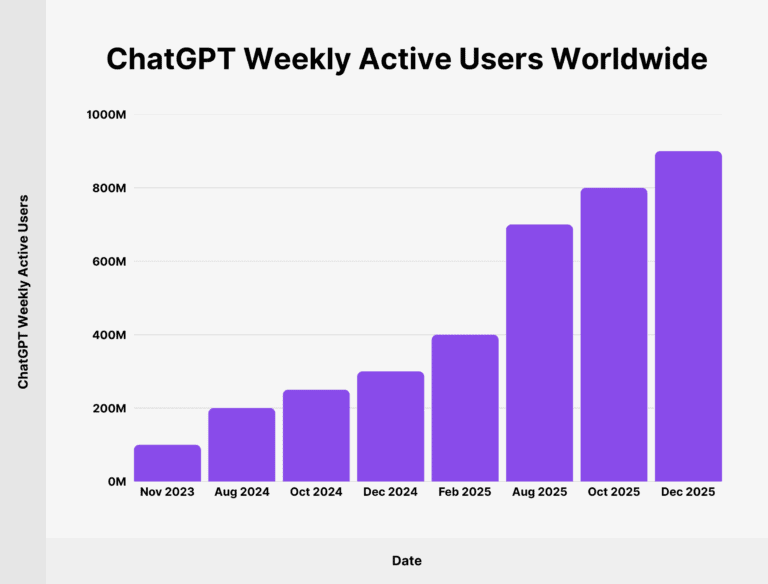
Figure 1: Changes over time in the number of ChatGPT users, source: https://backlinko.com/chatgpt-stats
Unlike what happened with search engines, the generative AI ecosystem is not dominated by a single player: while ChatGPT is the most widely used tool, a wide range of other solutions coexist, many of which are specifically designed for the medical field.
The available data indicate that these tools have already achieved significant adoption in medical practice:
- A 2023 study by the Healthcare Data Institute in France shows that 41.47% of doctors in France use AI at least once a month
- Another U.S. study published by the AMA (American Medical Association) on March 12, 2026, shows that more than 80% of American doctors are using artificial intelligence tools in 2026, compared to of 38% in 2023
- As for patients, OpenAI reports that of its 800 million regular users, 1 in 4 (25%) submits a health-related prompt every week
And beyond mere use, early clinical studies show that recommendations from large language models (LLMs) actually influence treatment decisions: a study published in *Nature Medicine* in 2025 involving 50 American doctors found that GPT-4’s recommendations led doctors to revise their initial clinical decisions, resulting in a measurable improvement in diagnostic accuracy.
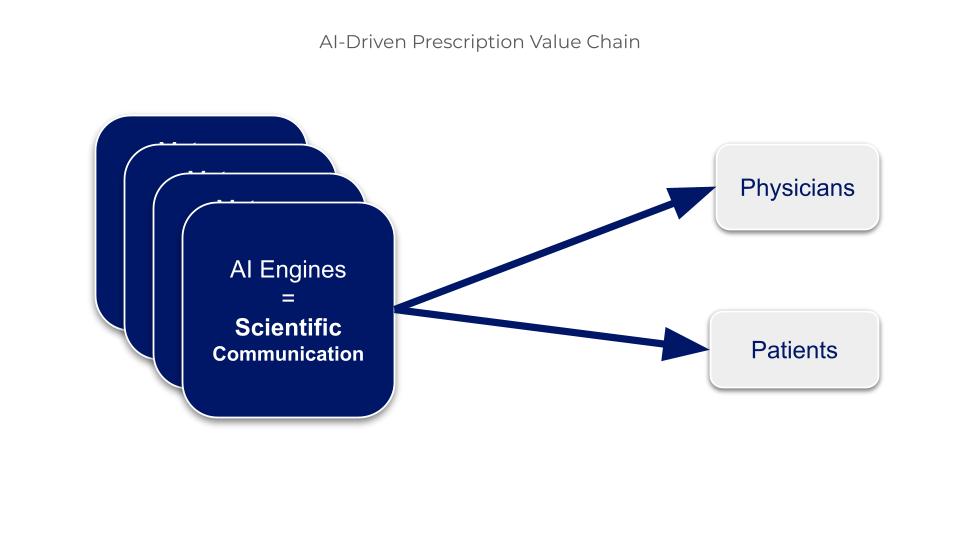
Figure 2. Artificial Intelligence: The Primary Influencer and Advisor for Doctors and Patients
Until now, there has been a lot of talk about the influence of Dr. Google, but Dr. AI has become the most powerful influencer in the entire healthcare ecosystem. This shift could be seen as a simple change in interface; in reality, it is a fundamentally different change: whereas Google merely lists web pages without assessing their reliability, AI tools synthesize and prioritize information by giving preference to validated scientific literature; for a medical question, Dr. AI responds as a doctor, not as a search engine.
-
Dr. IA's prescribing authority is directly linked to medical research publications containing longitudinal data
Tools from GEO (Generative Engine Optimization), which are used for optimization for generative engines, help identify the main sources that contribute to generating answers to users’ questions. There are also tools specific to the pharmaceutical industry, such as PharmaGEO, developed by the company Aikka.
This type of tool makes it possible to identify, for each artificial intelligence engine, the key medical research papers that examine all molecules and drugs.
In other words, the scientific community of doctors who publish in scientific journals directly feeds artificial intelligence engines and, consequently, all doctors and patients.
But to produce a high-quality medical research article, one needs large volumes of structured, homogeneous, longitudinal data specific to a particular therapeutic area. And the best sources of such data are, without a doubt, digital health applications that track patients throughout their medical treatment.
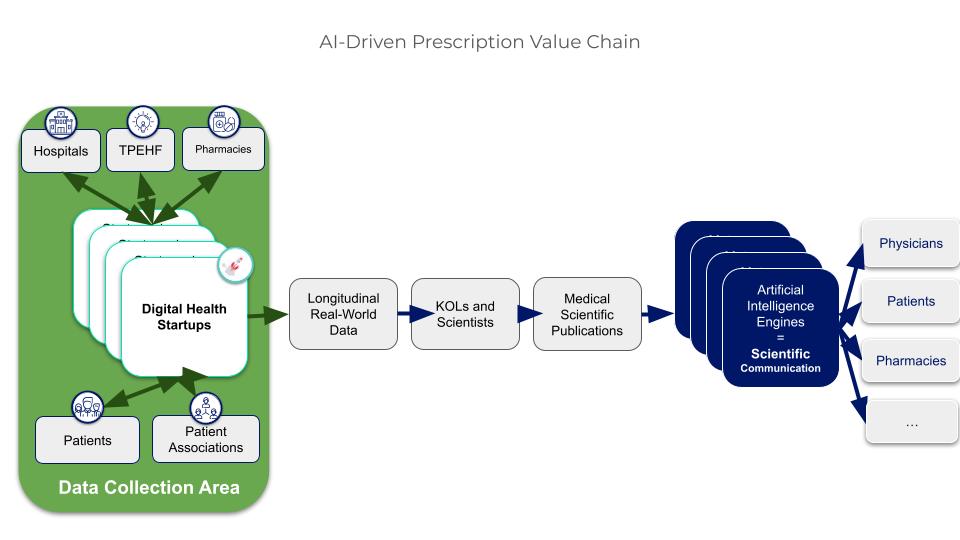
Figure 3. Digital health startups are the best sources of real-world data for scientific articles
Indeed, the literature shows that the value of digital data is primarily recognized when that data is linked to a clearly defined condition or indication, clinical outcomes, and treatment decisions—which is typically the case with solutions targeted at specific therapeutic areas rather than general health or wellness applications.
To reach patients on the necessary scale, these startups must necessarily be supported by key players in the healthcare system: doctors, particularly those in hospitals, as well as patient organizations and pharmacies.
We should also mention the new and unique role of Third-Party Experimentation Sites, a recent initiative by the French government to facilitate experiments within hospitals with digital health startups. There are currently 37 TLEs in France, and their future remains uncertain, even though they play a fundamental role in this new value creation.
The value of these startups—and their data—is directly proportional to their ability to scale: the greater the volume of data collected, the greater their scientific and strategic value.
However, we sometimes see a proliferation of startups in the same specialty or therapeutic area, which often creates confusion when choosing between these options. We will now see that this phase is temporary: for each therapeutic area considered separately—cardiology, diabetes care, oncology, etc.—we can predict that only a small number of players will emerge as leaders. However, this consolidation will not occur as quickly or as uniformly as in other digital markets: highly variable regulations across countries, the resistance of hospital institutions to becoming dependent on private players, and the natural fragmentation by specialty are all obstacles that will slow down the process. This is precisely why pharmaceutical companies seeking to establish a foothold have no interest in waiting, and why open collaborative models—which we will examine in Section 6—will be the sine qua non for the success of these acquisitions.
A paradoxical situation is emerging: digital health startups are at the heart of the new prescription value chain, yet they remain economically fragile. To understand how this paradox might be resolved, the history of ERP systems offers a striking insight.
-
Digital health startups are the ERP systems of the healthcare sector
Digital health has long been recognized by major international organizations as a key driver of transformation in our health systems : the World Health Assembly unanimously adopted a resolution on digital health, and the WHO has defined a global strategy aimed at making health systems people-centered and digitally enabled.
Nevertheless, these same startups are struggling to find a viable business model that will allow them to grow and build their market value.
In fact, today in France, the main business models for digital health startups are:
- direct sales to doctors, hospitals, and/or patients
- Reimbursement for telemedicine services by Social Security
- Reimbursement for digital therapies by Social Security
Reimbursement for telemedicine solutions and digital therapies in France, introduced after Germany’s, once seemed to provide a framework that would enable the rise of digital health. Today, however, it is clear that this is not the case; obtaining reimbursement is both a lengthy and complicated process, and it does not guarantee commercial success.
That is why the emergence of this new model of AI-powered prescribing could quickly become a real catalyst for the development of digital health.
The analogy with ERP (Enterprise Resource Planning) is illuminating. This software, deployed in all companies starting in the late 1990s, made it possible for each organization to identify the key data to collect and then create a centralized dashboard providing a comprehensive view of its operations; this is how companies were able to steer their transformation.
This analogy can be used to better understand how digital health solutions—by defining the data for each therapeutic area and then collecting that data across all stages of the care pathway—will provide a comprehensive view based on relevant and consistent data.
Ultimately, if this data is as crucial to businesses as ERP systems, then whoever controls these startups will control a portion of the market influence in the future. This is precisely what will trigger a race to acquire them.
-
The Decline in Digital Health Startups: A Windfall for Pharmaceutical Companies
Experience with digital markets reveals a recurring pattern. An initial exploration phase sees the emergence of numerous competing solutions: users generally adopt the first available solution and show some reluctance to expand their use of it. Gradually, a selection process takes place: the least effective solutions disappear, while those with the widest adoption attract new users, drawn by network effects. This process accelerates market concentration toward an oligopolistic structure, where a small number of players share the bulk of the market.
This oligopolistic market structure puts startups in a position of strength: with a significant market share, they are able to impose favorable contractual terms on pharmaceutical companies seeking to enter the market, which helps increase their valuation. Faced with this balance of power, pharmaceutical companies have two strategies to choose from to secure their partnerships: direct acquisition or taking an equity stake.
Several initiatives already illustrate this trend:
- Roche Laboratories acquired Flatiron Health in 2018 for $1.9 billion
- Sanofi's partnership and investment in Aetion in 2019
Furthermore, any pharmaceutical company that acquires one of these startups would gain exclusive control over a rare and strategic resource. As the number of players shrinks, acquiring these platforms will create a significant windfall effect: laboratories in a position to control access to this data could deny it to their competitors, thereby establishing a lasting competitive advantage at the expense of those who have delayed their decision.
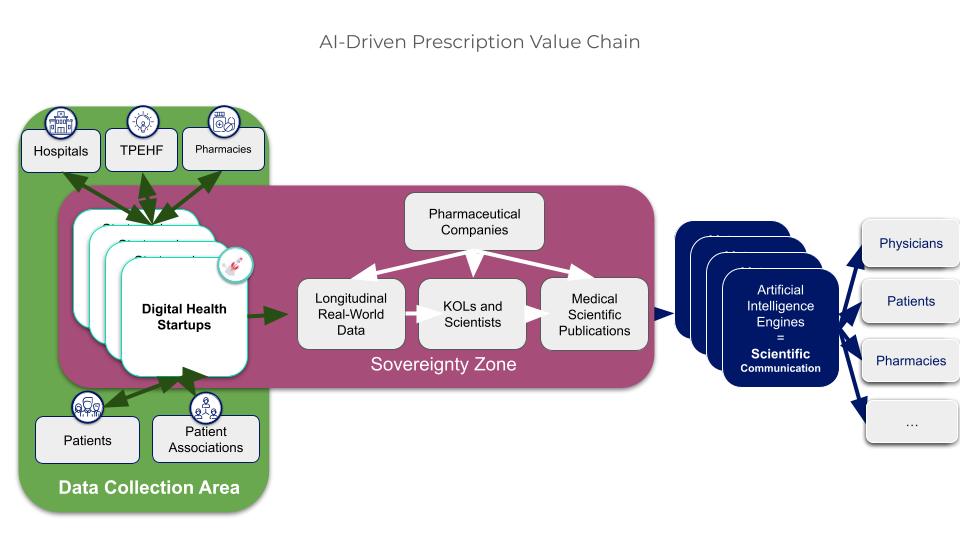 Figure 4. The acquisition of digital health startups creates a windfall for pharmaceutical companies
Figure 4. The acquisition of digital health startups creates a windfall for pharmaceutical companies
However, this acquisition strategy is not without risks: a pharmaceutical company’s control over a startup creates potential tensions with the ecosystem on which the startup’s value depends.
-
Open collaborative architecture: a prerequisite for the long-term viability of acquisitions
In fact, the interests of pharmaceutical companies may not be fully aligned with all the components of the ecosystem that drive the startup’s value and enable the production of this high-quality data.
Real tensions can arise between those who control the startup and the ecosystem surrounding it, which can lead to conflict and the startup being rejected.
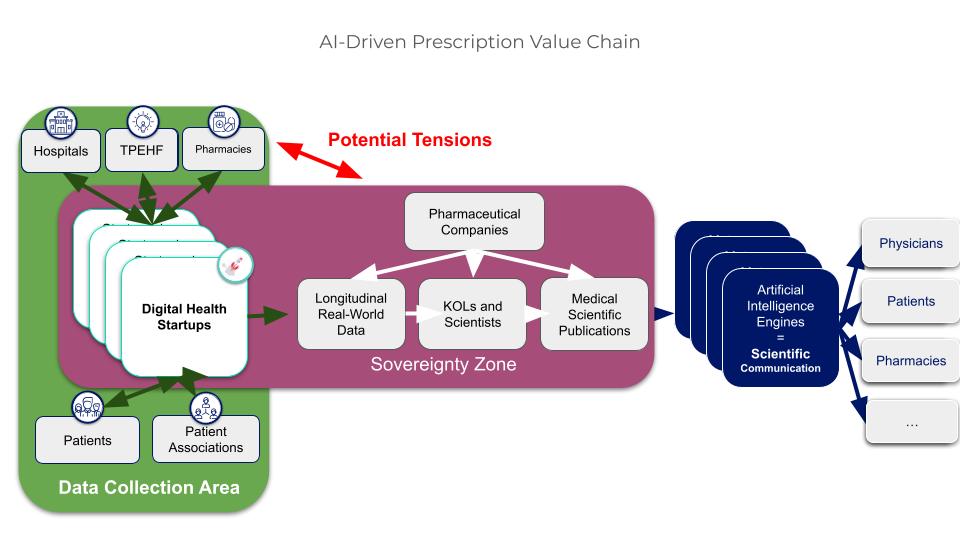
Figure 5. Illustration of potential tensions between pharmaceutical companies and the healthcare ecosystem
For such an acquisition to be sustainable, the pharmaceutical company controlling the startup must not act as a predator toward the ecosystem that constitutes its value. Three conditions are essential: first, multi-stakeholder governance that aligns everyone’s interests around shared objectives; second, a value-sharing model that benefits all contributors—doctors, hospitals, patient associations, and pharmacies—without whom the data cannot be produced; third, the capacity for concrete experimentation in the field, which validates the usefulness of solutions and generates real proof of value. These three conditions define what can be called an open collaborative architecture.
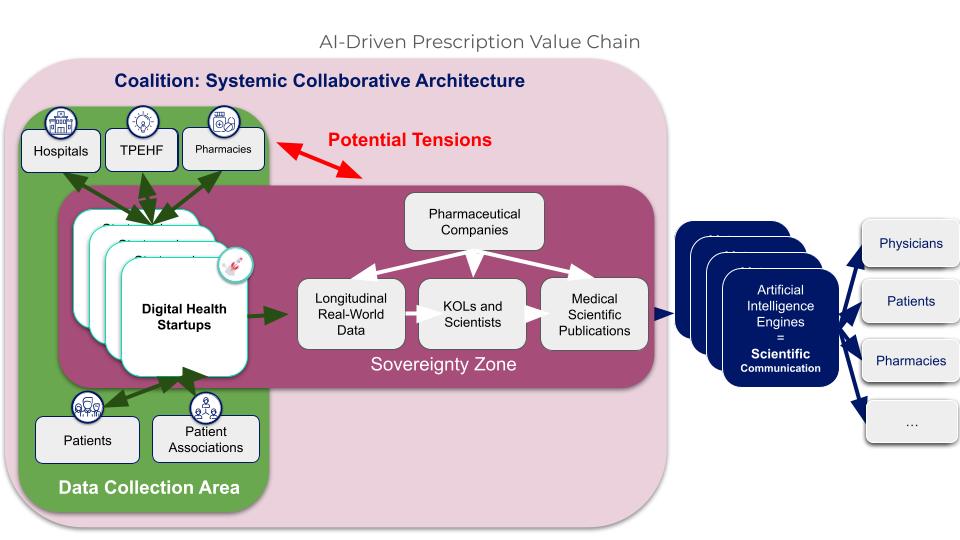
Figure 6. We need to build coalitions that align the interests of stakeholders in the healthcare ecosystem
NextGen Coalition, co-founded and operated by Digital Pharma Lab since 2020, is now the benchmark model for this type of architecture in France. In five years, it has brought together more than 40 partner institutions, hospitals, pharmaceutical companies, startups, and institutions, under a collective governance structure, raising €3.5 million and supporting more than 35 innovative projects covering digital care pathways, telemonitoring, and AI tools for clinicians, with 100,000 patients already benefiting from the solutions tested within this framework.
In the pharmaceutical sector, The IDEO Coalition, also led by Digital Pharma Lab, applies the same principle to a different ecosystem. It brings together healthcare manufacturers, pharmacy groups, distributors, and digital experts around a common goal: to support pharmacies in integrating digital technology, as the role of the pharmacist rapidly diversifies to include vaccination, screening, and therapeutic support. Each year, a call for projects—developed collaboratively with all members over six months of collective work—selects the most promising solutions, ensuring that the real needs of pharmacies and patients are at the heart of the initiative. The first call for projects received 42 applications, 11 of which were presented to the Coalition, and 4 projects were selected and are currently being finalized and rolled out.
These two examples illustrate a key lesson: the value of a coalition cannot be imposed; it must be built patiently, through proven results. What makes these models robust is precisely that they are not controlled by a single actor: they work because every stakeholder has a real and measurable stake in them. It is this model that pharmaceutical companies acquiring digital health startups will need to replicate and institutionalize, lest they destroy the very value they seek to capture.
-
In conclusion, AI will create a virtuous cycle that supports digital health startups and fosters genuine collaborative systems for the benefit of all
Artificial intelligence is fundamentally reshaping the pharmaceutical prescribing model by providing both doctors and patients with answers grounded in validated scientific literature. In this new landscape, digital health startups that generate large volumes of data in large volumes—structured, homogeneous, real-time, longitudinal, and specific to a therapeutic area— will enable the production of scientific publications grounded in a comprehensive, quantitative, and representative view of the therapeutic journey.
To safeguard their independence and strengthen their competitive edge, pharmaceutical companies would be well advised to acquire or gain control of these startups. To do so, however, they will need to build truly open collaborative frameworks capable of aligning the interests of all ecosystem stakeholders around this new value chain—a prerequisite for sustainable systemic change that benefits the entire sector.
Bibliography
Gagnon, M. A., & Lexchin, J. (2010). Information from pharmaceutical companies and the quality, quantity, and cost of physicians’ prescribing: A systematic review. PLOS Medicine, 7(10), e1000352. https://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1000352
Romanow, K. (August 18, 2015). Crossing the threshold: More than half of physicians restrict access to sales reps. PR Newswire. https://www.prnewswire.com/news-releases/crossing-the-threshold-more-than-half-of-physicians-restrict-access-to-sales-reps-300136525.html
PR Newswire. (July 14, 2016). As doctors continue to turn away pharmaceutical sales representatives, do digital communications offer a better solution? PR Newswire. https://www.prnewswire.com/news-releases/as-doctors-keep-closing-doors-on-pharma-reps-do-digital-communications-provide-a-better-solution-300317132.html
Healthcare Data Institute. (2023). The French, Healthcare Professionals, and Artificial Intelligence. https://healthcaredatainstitute.com/wp-content/uploads/2024/01/etudes-hdi-day-2023_compressed_compressed-1.pdf
American Medical Association. (March 12, 2026). More than 80% of physicians use AI professionally: AMA survey.
https://www.ama-assn.org/practice-management/digital-health/more-80-physicians-use-ai-professionally-ama-survey
Fierce Healthcare. (2024). 40 million people use ChatGPT to answer healthcare questions, OpenAI says. https://www.fiercehealthcare.com/ai-and-machine-learning/40m-people-use-chatgpt-answer-healthcare-questions-openai-says
Semigran, H. L., Linder, J. A., Gidengil, C., & Mehrotra, A. (2017). The effect of Dr. Google on doctor–patient encounters in primary care: A quantitative, observational, cross-sectional study. BMC Family Practice. https://pmc.ncbi.nlm.nih.gov/articles/PMC6169945
Staudenmayer, K. L., et al. (2025). Modification of physicians’ clinical decisions and assessment of bias in a randomized controlled trial of AI assistance. Communications Medicine (Nature). https://www.nature.com/articles/s43856-025-00781-2
Wikimedia Foundation. (2024). Optimization for Generative Engines. Wikipedia. https://fr.wikipedia.org/wiki/Optimisation_pour_les_moteurs_g%C3%A9n%C3%A9ratifs
Aikka. (n.d.). PharmaGEO. https://www.aikka.ai/
Neves, A. L., et al. (2023). Digital health technology for real-world clinical outcome measurement using patient-generated data: A systematic scoping review. Journal of Medical Internet Research, 25, e46992. https://www.jmir.org/2023/1/e46992/
GNIUS – Digital Health Platform. (2023). Third Places for Health Experimentation (TLE) – Stakeholder Profile. https://gnius.esante.gouv.fr/fr/acteurs/fiches-acteur/tiers-lieux-dexperimentation-en-sante-tle
GNIUS – Digital Health Platform. (2023). Third Places for Health Experimentation (TLE) – national programs. https://gnius.esante.gouv.fr/fr/programmes-nationaux/tiers-lieux-dexperimentation
Sinsky, C., et al. (2018). Digital health and health systems of the future. National Library of Medicine. https://pmc.ncbi.nlm.nih.gov/articles/PMC6203414/
Wikimedia Foundation. (2024). Digital therapy. Wikipedia. https://fr.wikipedia.org/wiki/Th%C3%A9rapie_num%C3%A9rique
Wikimedia Foundation. (2024). Enterprise resource planning software. Wikipedia. https://fr.wikipedia.org/wiki/Progiciel_de_gestion_int%C3%A9gr%C3%A9
Digital Pharma Lab. (n.d.). NextGen Coalition. https://www.digitalpharmalab.com/en/nextgen-coalition/
Digital Pharma Lab. (n.d.). IDEO Coalition – Pharmacy Innovation. https://www.digitalpharmalab.com/coalition-ideo-innovation-officine/
Wikimedia Foundation. (2024). Large language model. Wikipedia. https://fr.wikipedia.org/wiki/Grand_mod%C3%A8le_de_langage
JUNE 26. 2025

MAY 05. 2025
